[ad_1]
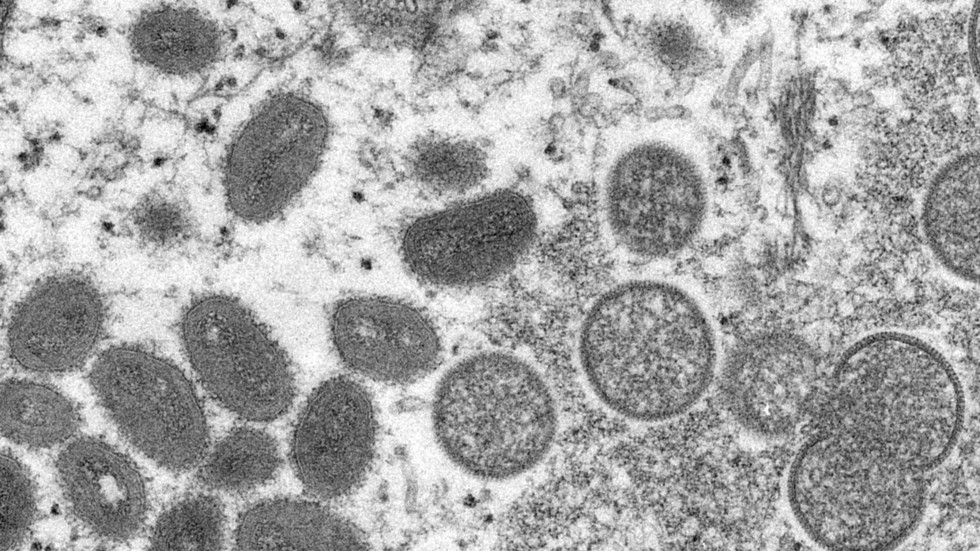
Officers have rushed to safe vaccines as circumstances of the uncommon monkeypox virus proceed to crop up within the US and Europe
The US well being authorities have signed a deal for $119 million in vaccine doses towards the monkeypox virus, after a Massachusetts man was identified with the uncommon however doubtlessly severe sickness earlier this week.
The Biomedical Superior Analysis and Growth Authority (BARDA) – a authorities company dedicated to combating pandemics and bioterrorism – signed the multi-million-dollar contract with Danish pharma agency Bavarian Nordic on Wednesday, the corporate introduced in an announcement.
The $119 million deal is one in a collection of contract choices which might in the end attain a complete worth of $299 million if exercised, in change for round 13 million freeze-dried doses of the Jynneos vaccine. It was initially created for smallpox, however was accepted to be used towards monkeypox by the Meals and Drug Administration (FDA) in 2019, simply months earlier than the primary circumstances of Covid-19 had been detected in China.
Preliminary deliveries for the Jynneos shot received’t come till 2023, the corporate mentioned, noting that the complete 13 million doses are anticipated to be prepared someday between 2024 and 2025 ought to BARDA agree to increase the contract.
The primary monkeypox case within the US was confirmed on Wednesday in a person who had lately traveled to Canada. Federal well being officers have since mentioned they’re monitoring six others after they got here into shut proximity to an contaminated traveler throughout a flight from Nigeria to the UK earlier this month, whereas one other doable case is being investigated by the New York Metropolis Well being Division.
A lot of suspected or confirmed infections have additionally been noticed in Britain, Canada, Spain, Portugal, Italy, and Sweden in current weeks. Australia has simply detected its first case.
On Thursday, Bavarian Nordic introduced that it had additionally reached a cope with an “undisclosed European nation” for a similar dual-use smallpox vaccine – although supplied underneath completely different branding – “in response to new circumstances of monkeypox.” The corporate didn’t elaborate on what number of doses could be procured or present an total price ticket for the contract.
Along with vaccines, the US authorities additionally moved to purchase up doses of tecovirimat, the usual antiviral therapy for monkeypox, with the Division of Protection signing a $7.5 million contract for the drug with American pharmaceutical agency SIGA Applied sciences final week.
An intravenous type of the identical antiviral obtained FDA approval for therapy of smallpox on Thursday, although SIGA mentioned the IV model was additionally “cited within the current US president’s finances request as getting used to deal with a affected person within the US with monkeypox.”
Whereas uncommon, monkeypox has been detected within the US earlier than, with a Texas resident changing into hospitalized from the virus final summer season after touring to West Africa, the place the pathogen is endemic. In 2003, greater than 70 circumstances had been confirmed within the US, marking the primary outbreak seen exterior of Africa, based on the World Well being Group.
Preliminary signs embrace fever, head and muscle aches, swollen lymph nodes, chills, and exhaustion, and although most infections resolve with out severe sickness, the virus is deadly in a small proportion of circumstances.
READ MORE:
Uncommon illness spreads in Europe
You may share this story on social media:
[ad_2]
Source link